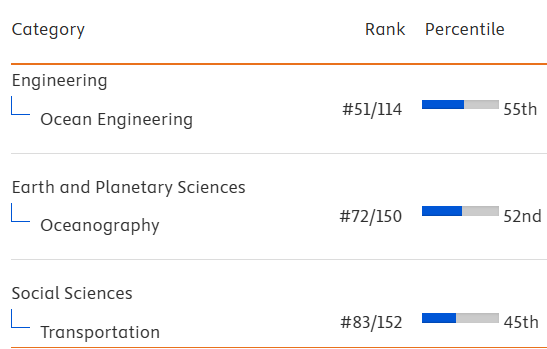
Behavior response, growth, and structure of the eye retinal layer in juvenile Seabass, Lates calcarifer: Light-induced changes
DOI:
https://doi.org/10.33175/mtr.2023.257541Keywords:
Aquaculture, Red light, Photoreceptor cell, Seabass, WavelengthsAbstract
Light is a key environmental factor that is strongly related to fish activity and behavior. Exposure to certain wavelengths is known to affect growth performance and survival of teleost larvae and juveniles. Our study aimed to determine the effects of exposure to three light sources [red (710 nm), blue (453 nm) and green (510 nm)] on growth performance, behavioral response, visibility (time to eat, in minutes), and the structure of the retinal layer in juvenile [approx. 4.3 cm in total length (N = 30)] seabass (Lates calcarifer). Fish were acclimated to the three light conditions for four weeks prior to data collection. Behavioral responses, including schooling organization, swimming speed, time to consume prey, and feeding response, were all improved in juveniles reared under red light (710 nm). Preliminary histological observations revealed that fish from the red-light environment have the thickest retina layers and the highest density of photoreceptors. Our data suggest that red light is useful in the aquaculture of juvenile L. calcarifer.
------------------------------------------------------------------------------
Cite this article:
APA Style:
Kaewpranee, S., Kettratad, J., Angsujinda, K., Senarat, S., Charoenphon, N., Plumley, F.G., Sayoh, I., & Kaneko, G. (2023). Behavior response, growth, and structure of the eye retinal layer in juvenile Seabass, Lates calcarifer: Light-induced changes. Maritime Technology and Research, 5(1), 257541. https://doi.org/10.33175/mtr.2023.257541
MDPI Style:
Kaewpranee, S.; Kettratad, J.; Angsujinda, K.; Senarat, S.; Charoenphon, N.; Plumley, F.G.; Sayoh, I.; Kaneko, G. Behavior response, growth, and structure of the eye retinal layer in juvenile Seabass, Lates calcarifer: Light-induced changes. Marit. Technol. Res. 2023, 5(1), 257541. https://doi.org/10.33175/mtr.2023.257541
Vancouver Style:
Kaewpranee S, Kettratad J, Angsujinda K, Senarat S, Charoenphon N, Plumley FG, Sayoh I, Kaneko G. (2023). Behavior response, growth, and structure of the eye retinal layer in juvenile Seabass, Lates calcarifer: Light-induced changes. Marit. Technol. Res. 5(1): 257541. https://doi.org/10.33175/mtr.2023.257541
------------------------------------------------------------------------------
Highlights
- Behavioral responses of seabass (Lates calcarifer), including schooling organization, swimming speed, time to consume prey, and feeding response, were all improved in juveniles reared under red light (710 nm).
- Histological observations revealed that fish from the red-light environment have the thickest retina layers and highest density of photoreceptors.
- Red light is optimum for cultivation of calcarifer juveniles, which might stimulate motivation for feeding activity and improvement for growth rate of its aquaculture and farms.
References
Ahmad, A. M., Shoman, H. M., El-Deeb, R. M., Abdelhafez, H. M., & Samei, E. A. (2017). Comparative studies on the histology of eye retina in some Nile fishes with different dial activities. Egyptian Journal of Hospital Medicine, 68, 815-823. https://doi.org/10.12816/0038179
Ballagh, D. A., Pankhurst, P. M., & Fielder, D. S. (2008). Photoperiod and feeding interval requirements of juvenile mulloway, Argyrosomus japonicus. Aquaculture, 277, 52-57. Https://doi.org/10.1016/j.aquaculture.2008.02.025
Begg, K., & Pankhurst, N. W. (2004). Endocrine and metabolic responses to stress in a laboratory population of the tropical damselfish Acanthochromis polyacanthus. Journal of Fish Biology, 64, 133-145. https://doi.org/ 10.1111/j.1095-8649.2004.00290.x
Blaxter, J. H. S. (1968). Light intensity, vision and feeding in young plaice. Journal of Experimental Marine Biology and Ecology, 2, 293-307. https://doi.org/10.1016/0022-0981(68)90021-X
Boeuf, G., & Bail, P. Y. L. (1999). Does light have an influence on fish growth? Aquaculture, 177, 129-152. https://doi.org/10.1016/S0044-8486(99)00074-5
Cuvier-Péres, A., Jourdan, S., Fontaine, P., & Kestemont, P. (2001). Effects of light intensity on animal husbandry and digestive enzyme activities in sea bass Dicentrachus labrax post-larvae. Aquaculture, 202, 317-328. https://doi.org/ 10.1016/S0044-8486(01)00781-5
Department of Fisheries. (2017). Statistical analysis of estuarine fish, 2017. Department of Fisheries Science. Retrieved from https://www.fisheries.go.th/strategy-stat/themeWeb/books/2559/1/yearbook_2559.pdf
De Jesus-Ayson, E. G., & Ayson, F. G. (2014). Reproductive biology of the Asian seabass, Lates calcarifer (pp. 67-76). In Jerry, D. R. (Ed.). Biology and Culture of Asian Seabass Lates calcarifer. CRC Press, Boca Raton. https://doi.org/10.1201/b15974-6
Donatti, L., & Fanta, E. (1999). Morphology of the retina in the freshwater fish Metynnis roosevelti Eigenmann (Characidae, Serrasalminae) and the effects of monochromatic red light. Revista Brasileira de Zoologia, 16, 151-173. https://doi.org/10.1590/S0101-81751999000100011
Downing, G., & Litvak, M. K. (1999). The effect of photoperiod, tank colour and light intensity on growth of larval haddock. Aquaculture International, 7, 369-382. https://doi.org/10.1023/A:1009204909992
Fielder, D. S., Bardsley, W. J., Allan, G. L., Fielder, D. S., & Pankhurst, P. M. (2002). Effect of photoperiod on growth and survival of snapper Pagrus auratus larvae. Aquaculture, 211, 135-150. https://doi.org/10.1016/S0044-8486(02)00006-6
Genten, F., Terwinghe, E., & Danguy, A. (2009). Atlas of fish histology (pp. 1-138). Science Publishers. https://doi.org/10.1201/9780367803599
Glencross, B., Wade, N., & Morton, M. K. (2013). Lates calcarifer nutrition and feeding practices (pp. 1-326). Taylor and Francis, Boca Raton.
Glencross, B. (2006). Nutritional requirements: Carnivorous fin-fish. Aquaculture nutrition master class (pp. 1-40). Bangkok, Thailand: Asian Institute of Technology.
Guiguen, Y., Cauty C., Fostier, A., Jacques F., & Jalabert, B. (1994). Reproductive cycle and sex inversion of the Seabass, Lates calcarifer, reared in sea cages in French Polynesia: Histological and morphometric description. Environmental Biology of Fishes, 39, 231-247. https://doi.org/10.1007/BF00005126
Gunnarsson, S., Imsland, A. K., Siikavuopiom, S. I., Árnason, J., Gústavsson, A., & Thorarensen A. (2012). Enhanced growth of farmed Artic charr (Salvelinus alpinus) following a short day photoperiod. Aquaculture, 350-353, 75-81. https://doi.org/10.1016/j.aquaculture.2012.04.014
Heerhartz, S. M., & Toft, J. D. (2015). Movement patterns and feeding behavior of juvenile salmon (Oncorhynchus spp.) along armored and unarmored estuarine shorelines. Environmental Biology of Fishes, 98, 1501-1511. https://doi.org/10.1007/s10641-015-0377-5
Howell, A., Berlinsky, D. L., & Bradley, T. M. (2003). The effect of photoperiod manipulation in the reproduction of black sea bass, Centropristis striata. Aquaculture, 218, 651-669. https://doi.org/10.1016/S0044-8486(02)00343-5
Hunt, D. E., Rawlinson, N. J. F., Thomas, G. A., & Cobcroft, J. M. (2015). Investigating photoreceptor densities, potential visual acuity, and cone mosaics of shallow water, temperate fish species. Vision Research, 111, 13-21. https://doi.org/10.1016/j.visres.2015.03.017
IBM Crop. (2015). IBM SPSS Statistics for Windows, Version 23.0. Armonk, NY: IBM Crop.
Imsland, A. K., Foss, A., Stefansson, S. O., Mayer, I., Norberg, B., Roth, B., & Jenssen, M. D. (2006). Growth, feed conversion efficiency and growth heterogeneity in Atlantic halibut (Hippoglossus hippoglossus) reared at three different photoperiods. Aquaculture Research, 37, 1099-1106. https://doi.org/10.1111/j.1365-2109.2006.01533.x
Job, S. D., & Bellwood, D. R. (2007). Light sensitivity in larval fishes: Implications for vertical zonation in the pelagic zone. Limnology and Oceanography, 45, 362-371. https://doi.org/10.4319/lo.2000.45.2.0362
Karakatsouli, N., Papoutsoglou, S. E., Pizzonia, G., Tsatsosa, G., Tsopelakos, A., Chadio, S., Kalogiannis, D., Dalla, C., Polissidis, A., & Papadopoulou-Daifotic, Z. (2007). Effects of light spectrum on growth and physiological status of gilthead seabream Sparus aurata and rainbow trout Onchorhynchus mykiss reared under recirculating system conditions. Aquacultural Engineering, 36, 302-309. https://doi.org/10.1016/j.aquaeng.2007.01.005
Kawamura, G., Bagarinao T. U., & Lim, L. S. (2015). Fish behaviour and aquaculture (pp. 68-106). In Mustafa, S., & Shapawi, R. (Eds.). Aquaculture Ecosystems: Adaptability and Sustainability. Wiley-Blackwell, Oxford. https://doi.org/10.1002/9781118778531.ch3
Kawamura, G., & Ishida, K. (1985). Changes in sense organ morphology and behaviour with growth in the flounder Paralichthys olivaceus. Bulletin of the Japanese Society of Scientific Fisheries, 51, 155-165. https://doi.org/10.2331/suisan.51.155
Kawamura, G., Tsuda, R. Kumai, H., & Ohashi, S. (1984). The visual cell morphology of Pagrus major and its adaptive changes with shift from pelagic to benthic habitats. Bulletin of the Japanese Society of Scientific Fisheries, 50, 1975-1980. https://doi.org/10.2331/suisan.50.1975
Kim, H. H., Goins, G. D., Wheeler, R. M., & Sager, J. C. (2004). Green-light supplementation for enhanced lettuce growth under red-and blue-light-emitting diodes. HortScience, 39, 1617-1622. https://doi.org/10.21273/HORTSCI.39.7.1617
Kobayashi, H. (1962). A comparative study on electroretinogram in fish, with special reference to ecological aspects. Journal of Shimonoseki University of Fisheries, 11, 407-538.
Li, N., Zhou, J., Wang, H., Wang, C., Mu, C., Shi, C., & Liu L. (2020). Effects of light intensity on growth performance, biochemical composition, fatty acid composition and energy metabolism of Scylla paramamosain during indoor overwintering. Aquaculture Reports, 18, 100443. https://doi.org/10.1016/j.aqrep.2020.100443
Lythgoe, J. N., & Partridge, J. C. (1989). Visual pigment and the acquisition of visual information. Journal of Experimental Biology, 146, 1-20. https://doi.org/10.1242/jeb.146.1.1
Marchesan, M., Spoto, M., Verginella, L. & Ferrero, E.A. (2005). Behavioural effects of artificial light on fish species of commercial interest. Fisheries Research, 73, 171-185. https://doi.org/10.1016/j.fishres.2004.12.009
Martínez-Cárdenas, L., & Purser, G. J. (2011). Effect of stocking density and photoperiod on growth and survival in cultured early juvenile pot-bellied seahorses Hippocampus abdominalis Lesson, 1827. Aquaculture Research, 43, 1536-1549.
https://doi.org/10.1111/j.1365-2109.2011.02958.x
Matsuoka, M. (1999). Histological characteristics and development of the retina in the Japanese sardine Sardinops melanostictus. Fisheries Science, 65, 224-229. https://doi.org/10.2331/fishsci.65.224
Naas, K., Huse, I., & Iglesias, J. (1996). Illumination in first feeding tanks for marine fish larvae. Aquacultural Engineering, 15, 291-300. https://doi.org/10.1016/0144-8609(95)00019-4
Parr, A. E. (1927). A contribution to the theoretical analyses of the schooling behavior of fishes. Occasional papers of the Bingham Oceanographic Collection, 1, 1-32.
Parr, A. E. (1931). Sex dimorphism and schooling behavior among fishes. The American Naturalist, 65, 173-180. https://doi.org/10.1086/280359
Pillay, T. V. R., & Kutty, M. N. (2005). Aquaculture principles and practices (pp. 1-640). Blackwell, London.
Presnell, J. K., & Schreibman, M. P. (2013). Humason’s Animal Tissue Techniques (pp. 1-600). 5th eds. Johns Hopkins University Press, Baltimore.
Ruchin, A. B. (2004). Influence of colored light on growth rate of juveniles of fish. Fish Physiology and Biochemistry, 30, 175-178. https://doi.org/10.1007/s10695-005-1263-4
Suvarna, K. S., & Layton, J. D. (2013). Bancroft Bancroft’s Theory and Practice of Histological Techniques (pp. 1-573). 7th ed. Elsevier, Canada.
Trippel, E. A., & Neil, S. R. E. (2002). Effects of photoperiod and light intensity on growth and activity of juvenile haddock (Melanogrammus aeglefinus). Aquaculture, 217, 633-645. https://doi.org/10.1016/S0044-8486(02)00198-9
Ulmann, J. F. P., Gallagher, T., Hart, N. S., Barnes, A. C., Smullen, R. P., Collin, S. P., & Temple, S. E. (2011). Tank color increase growth and alters color preference and spectral sensitivity in barramundi (Lates calcarifer). Aquaculture, 32, 235-240. https://doi.org/10.1016/j.aquaculture.2011.10.005
Utne-Palm, A. C., Breen, M., Løkkeborg, S., & Humborstad, O. B. (2018). Behavioral response of krill and cod to artificial light in laboratory experiments. PLoS One, 13, e0190918. https://doi.org/10.1371/journal.pone.0190918
Volpato, G. L., Bovi, T. S., Freitas, R. H. A., Silva, D. F., Delicio, H. C., Giaquinto, P. C., & Barreto, R. E. (2013). Red light stimulates feeding motivation in fish but does not improve growth. PLoS One, 8(3), e59134. https://doi.org/10.1371/journal.pone.0059134
Wilson, J. M., Bunte, R. M., & Carty, A. J., (2009). Evaluation of rapid cooling and tricaine methanesulfonate (MS222) as methods of euthanasia in zebrafish (Danio rerio). Journal of the American Association for Laboratory Animal Science, 48, 785-789.
Yoshimatsu, T., Schröder, C., Nevala N. E., Berens P., & Baden T. (2020). Fovea-like photoreceptor specializations underlie single UV cone driven prey-capture behavior in Zebrafish. Neuron, 107, 320-337. https://doi.org/10.1016/j.neuron.2020.04.021
Downloads
Published
Issue
Section
License
Copyright (c) 2022 Maritime Technology and Research

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Copyright: CC BY-NC-ND 4.0