การพัฒนาและการตรวจสอบความใช้ได้ของวิธีทดสอบในการวิเคราะห์สาร Demethylwedelolactoneและสาร Wedelolactone (in Eclipta prostrata) ด้วยเทคนิค HPLC – PDA
Main Article Content
บทคัดย่อ
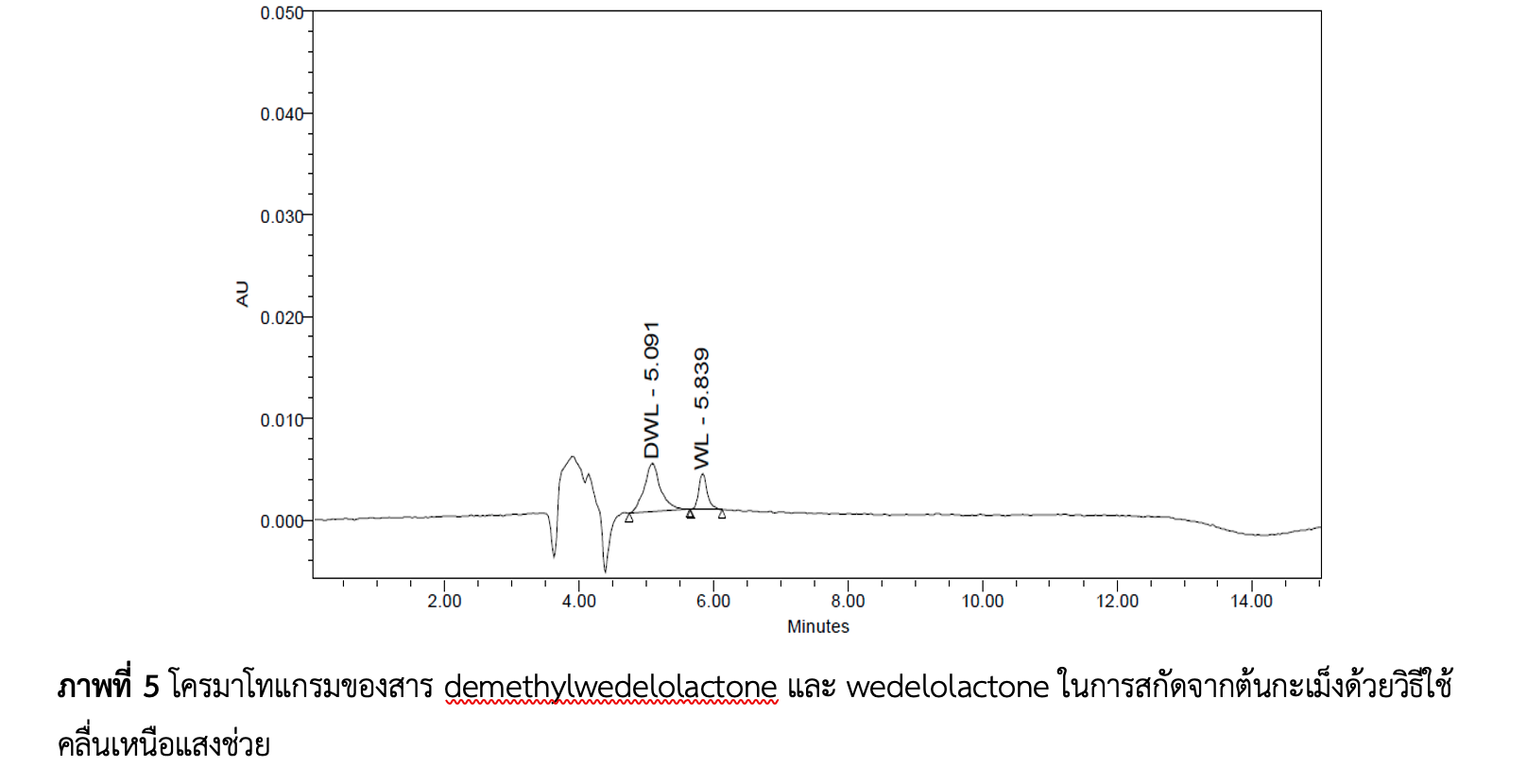
งานวิจัยนี้มีวัตถุประสงค์เพื่อพัฒนาและตรวจสอบความถูกต้องของวิธีวิเคราะห์สำหรับสารดีเมทิลวีดีโลแลคโตน และสารวีดีโลแลคโตน ด้วยเทคนิค HPLC – PDA ของสารสกัดจากต้นกะเม็งด้วยวิธีการสกัดที่ต่างกัน ผลการทดลองเลือกสภาวะที่เหมาะสมสำหรับการแยกสารดีเมทิลวีดีโลแลคโตน และสารวีดีโลแลคโตน คือ เฟสเคลื่อนที่ที่มีส่วนผสมของเมทานอลและสารละลายกรดฟอร์มิก 0.05% (โดยปริมาตร) อัตราส่วน 95:5 (โดยปริมาตร) กำหนดอัตราการไหลเท่ากับ 0.6 มิลลิลิตรต่อนาที ใช้การชะแบบแกรเดียนท์ จากการตรวจความใช้ได้ของวิธีทดสอบในการวิเคราะห์สารดีเมทิลวีดีโลแลคโตน และสารวีดีโลแลคโตน พบว่ามีค่าขีดจำกัดต่ำสุดของการตรวจวัด เท่ากับ 0.41 µg L-1 และ 0.004 µg L-1 ตามลำดับ ขีดจำกัดต่ำสุดของการหาปริมาณ เท่ากับ 1.37 µg L-1 และ 0.013 µg L-1 ตามลำดับ ศึกษาความสัมพันธ์เชิงเส้นตรงของสารดีเมทิลวีดีโลแลคโตน พบว่ามีช่วงความเข้มข้น 5.0 – 25 µg L-1 มีค่าสัมประสิทธิ์สหสัมพันธ์ เท่ากับ 0.998 และสารวีดีโลแลคโตน มีช่วงความเข้มข้น 1.0 – 25 µg L-1 มีค่าสัมประสิทธิ์สหสัมพันธ์ เท่ากับ 0.999 ตรวจสอบความเที่ยงของวิธีวิเคราะห์ จากผลการทดลองซ้ำในวันเดียวกันโดยศึกษาร้อยละส่วนเบี่ยงเบนมาตรฐานสัมพัทธ์ สารดีเมทิลวีดีโลแลคโตนมีค่าอยู่ในช่วง 0.38 – 1.61 และสารวีดีโลแลคโตนอยู่ในช่วง 1.18 – 1.46 และผลการทดลองซ้ำในแต่ละวันของสารดีเมทิลวีดีโลแลคโตนอยู่ในช่วง 0.34 – 0.47 และสารวีดีโลแลคโตนอยู่ในช่วง 0.59 – 1.20 ตรวจสอบความแม่นของวิธีวิเคราะห์ โดยวิธี spiked placebo recovery method พบว่า ร้อยละการกลับคืนของสารดีเมทิลวีดีโลแลคโตนอยู่ในช่วง 89.21 – 97.00% และสารวีดีโลแลคโตนอยู่ในช่วง 100.52 – 100.81 และโดยวิธี standard addition method พบว่า ร้อยละการกลับคืนของสารดีเมทิลวีดีโลแลคโตนอยู่ในช่วง 103.01 – 104.93 และสารวีดีโลแลคโตนอยู่ในช่วง 89.69 – 105.94 อยู่ในเกณฑ์การยอมรับได้ทั้ง 2 วิธี ผลการศึกษาวิธีการสกัดสารดีเมทิลวีดีโลแลคโตน และสารวีดีโลแลคโตน จากต้นกะเม็งด้วยวิธีแช่หมัก วิธีใช้คลื่นเหนือเสียงช่วยและวิธีการต้ม พบสารดีเมทิลวีดีโลแลคโตน มีปริมาณที่ไม่แตกต่างกันอย่างมีนัยสำคัญทั้ง 3 วิธี ส่วนสารวีดีโลแลคโตน พบเฉพาะในวิธีแช่หมักและวิธีใช้คลื่นเหนือเสียงช่วย
Article Details

อนุญาตภายใต้เงื่อนไข Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
วารสารวิทยาศาสตร์และวิทยาศาสตร์ศึกษา (JSSE) เป็นผู้ถือลิสิทธิ์บทความทุกบทความที่เผยแพร่ใน JSSE นี้ ทั้งนี้ ผู้เขียนจะต้องส่งแบบโอนลิขสิทธิ์บทความฉบับที่มีรายมือชื่อของผู้เขียนหลักหรือผู้ที่ได้รับมอบอำนาจแทนผู้เขียนทุกนให้กับ JSSE ก่อนที่บทความจะมีการเผยแพร่ผ่านเว็บไซต์ของวารสาร
แบบโอนลิขสิทธิ์บทความ (Copyright Transfer Form)
ทางวารสาร JSSE ได้กำหนดให้มีการกรอกแบบโอนลิขสิทธิ์บทความให้ครบถ้วนและส่งมายังกองบรรณาธิการในข้อมูลเสริม (supplementary data) พร้อมกับนิพนธ์ต้นฉบับ (manuscript) ที่ส่งมาขอรับการตีพิมพ์ ทั้งนี้ ผู้เขียนหลัก (corresponding authors) หรือผู้รับมอบอำนาจ (ในฐานะตัวแทนของผู้เขียนทุกคน) สามารถดำเนินการโอนลิขสิทธิ์บทความแทนผู้เขียนทั้งหมดได้ ซึ่งสามารถอัพโหลดไฟล์บทความต้นฉบับ (Manuscript) และไฟล์แบบโอนลิขสิทธิ์บทความ (Copyright Transfer Form) ในเมนู “Upload Submission” ดังนี้
1. อัพโหลดไฟล์บทความต้นฉบับ (Manuscript) ในเมนูย่อย Article Component > Article Text
2. อัพโหลดไฟล์แบบโอนลิขสิทธิ์บทความ (Copyright Transfer Form) ในเมนูย่อย Article Component > Other
ดาวน์โหลด ไฟล์แบบโอนลิขสิทธิ์บทความ (Copyright Transfer Form)
เอกสารอ้างอิง
Aderogba, M.A., McGaw, L.J., Ogundaini, A.O. and Eloff, J.N. (2007). Antioxidant activity and cytotoxicity study of the flavonol glycosides from Bauhinia galpinii. Natural Product Research, 21, 591-599.
Akeyothinwong, K., Sakkayawong, N., Damrianant, S. and Nakbanpote, W. (2020). Antioxidant activity and bioactive compounds of Kameng (Eclipta prostrata Linn.) extracts (in Thai). Thai Journal of Science and Technology, 9(1), 45-57.
Chansuvarn, W. (2021). Method validation. Retrieved 15 February 2022 from https://web.rmutp.ac.th/woravith/?page_id=8366
Chumark, P., Khunawat, P., Sanvarinda, Y., Phornchirasilp, S., Morales, N.P., Phivthong-ngam, L., Ratanachamnong, P., Srisawat, S. and Pongrapeeporn, Ku.S. (2008). The in vitro and ex vivo antioxidant properties, hypolipidaemic and antiatherosclerotic activities of water extract of Moringa oleifera Lam. leaves. Journal of Ethnopharmacology, 116(3), 439-446.
Jaisin, Y. (2016). Ka-meng (in Thai). Thai Journal of Pharmacology, 38(2), 40-47.
Kumar, S. and Dhanani, T. (2013). Development and validation of a rapid high performance liquid chromatography – photodiode array detection method for estimation of a bioactive compound wedelolactone in extracts of Eclipta alba. Brazilian Journal of Pharmaceutical Sciences. 49(1), 57-63.
Lee, H. Y. (2017). Enhancement of skin anti – inflammatory activities of Eclipta prostrata l. from the ultrasonic extraction process. Applied Sciences, 7, 1227.
Mendes, F.N.P., Morais, S.M.D., Silva, M.G.V., Vieira, I.R., Rodrigues, P.A.S., Belizáriodiniz, D. and Vieira. Í.G.P. (2014). Determination of wedelolactone and demethylwedelolactone in Eclipta alba (l) hassk by hplc. International Journal of Pharmacy and Pharmaceutical Sciences, 6(10), 403-406.
Morel, L.J.F., Azevedo, B.C., Carmona, F., Contini, S.H.T., Teles, A.M., Ramalho, F.S., Bertoni, B.W., Franca, S.C., Borges, M.C. and Pereira, A.M.S. (2017). A standardized methanol extract of Eclipta prostrata (L.) L. (Asteraceae) reduces bronchial hyperresponsiveness and production of Th2 cytokines in a murine model of asthma. Journal of Ethnopharmacology, 198, 226-234.
Murali, B., Amit, A., Anand, M. S. and Samiulla, D. S. (2002). Estimation of wedelolactone and
demethylwedelolactone in Eclipta alba Hassk. By improved chromatograhic analysis. Journal of Natural Remedies, 2(1), 99-101.
Nawaz, H., Shad, M.A., Rehman, N., Andaleeb, H. and Ullah, N. (2020). Effect of solvent polarity on extraction yield and antioxidant properties of phytochemicals from bean (Phaseolus vulgaris) seeds. Brazilian Journal of Pharmaceutical Sciences, 56, e17129.
Phale, M.D., Hamrapurkar, P.D., Chachad, M.A., Patil, P.S. and Pawar, S.B. (2010). Precise and sensitive HPTLC method for quantitative estimation of wedelolactone in Eclipta alba hassk. Pharmacophore, 1(2), 103-111.
Sasidharan, S., Chen, Y., Saravanan, D., Sundram, K.M. and Latha, L.Y. (2011). Extraction, isolation and characterization of bioactive compounds from plant's extracts. African Journal of Traditional, Complementary and Alternative Medicines, 8(1), 1-10.
Siramon, P. and Weanukul, R. (2020). Using ultrasound-assisted solvent extraction of phenolic compounds from Curcuma aromatica Salisb Rhizomes (in Thai). Journal of Science and Technology, Ubon Ratchathani University, 22(1), 40-44.
Snyder, L.R., Kirkland J.J. and Glajch J.L. (1997). Appendix II: Properties of Solvents Used in HPLC. In book: Practical HPLC Method Development, Second Edition.